Atoms
An atom is a basic unit that makes up all matter. There are many different types of atom, each with its own name, mass and size. These are called chemical elements, such as hydrogen or gold. Atoms are very small!The exact size changes depending on the element: they range from 0.1 to 0.5 nanometres. One nanometer is around a hundred times smaller than the width of a human hair. This makes atoms almost impossible to see. Equations must be used to see the way they work and how they interact with other atoms.
Atoms combine to make molecules or particles: for example, two hydrogen atoms and one oxygen atom combine to make a water molecule, a form of a chemical reaction.
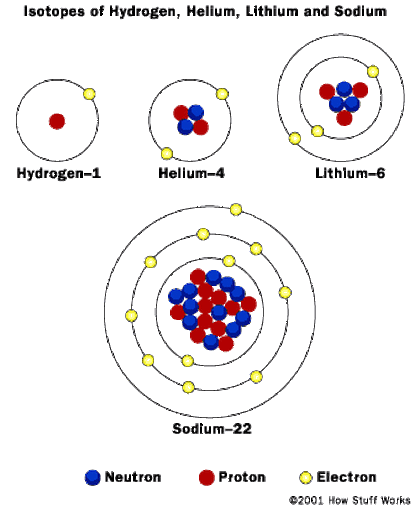
Atoms themselves are made up of three kinds of smaller particles, called protons, neutrons and electrons. The protons and neutrons are in the middle of the atom. They are called the nucleus. The nucleus is surrounded by a cloud of electrons with a negative charge which are bound to the nucleus by an electromagnetic force.
Below is a picture of how electrons are configured. This is further discussed in the electron configuration section of this website.
Atoms combine to make molecules or particles: for example, two hydrogen atoms and one oxygen atom combine to make a water molecule, a form of a chemical reaction.
Atoms themselves are made up of three kinds of smaller particles, called protons, neutrons and electrons. The protons and neutrons are in the middle of the atom. They are called the nucleus. The nucleus is surrounded by a cloud of electrons with a negative charge which are bound to the nucleus by an electromagnetic force.
Below is a picture of how electrons are configured. This is further discussed in the electron configuration section of this website.