Lab Report on Reaction Rate - Effect of Temperatures and Concentration
Hydrochloric Acid: HCl
Sodium Thiosulfate:Na2S2O3
AIM
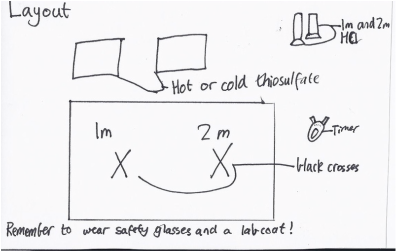
The aim of this experiment is to see how the reaction rate is effected by temperature, specifically heated and chilled sodium thiosulfate, and also concentration of hydrochloric acid, 1 mole and 2 moles to see how the speed of the chemical reaction is affected. The reason we are doing this is to investigate the Kinetic and Collision theories, and see how they work when we put them to the test. What we do in the experiment is heat and cool the sodium thiosulfate, and then add 1 mole and 2 mole concentrations of hydrochloric acid to heated and cooled sodium thiosulfate, and record the amount of time it takes to go white, or opaque. We know it is opaque because we have added a black cross underneath the beakers with the sodium thiosulfate. The time that we have recorded is called the 'reaction rate' The Collision theory explains how chemical reactions happen and why the rate of the reaction rates differ between different chemical reactions. It states that for a chemical reaction to occur, atoms, molecules, or chemical species have to collide in order to have a chemical reaction, but not all collisions bring chemical change. The Collision theory was proposed in 1916 by the German chemist Max Trautz, and British scientist William Lewis, in order to explain how chemical reactions and why rates differ for different reactions. In our experiment, we used different concentrations of hydrochloric acid, and in the Collision theory, lower concentrations have lower collisions, and higher concentrations have more collisions. That means that, theoretically, the higher concentrations of hydrochloric acid means that the 2 mole HCl will have more collisions, and that means that the reaction rate should be faster. Temperature is also a factor in the Collision theory. An increase in the temperature increases the average speed of the reactant molecules and the number or frequency of collisions. The Kinetic theory, states that the hotter the substance is, the faster the molecules move in it.
Sodium Thiosulfate:Na2S2O3
AIM
The aim of this experiment is to see how the reaction rate is effected by temperature, specifically heated and chilled sodium thiosulfate, and also concentration of hydrochloric acid, 1 mole and 2 moles to see how the speed of the chemical reaction is affected. The reason we are doing this is to investigate the Kinetic and Collision theories, and see how they work when we put them to the test. What we do in the experiment is heat and cool the sodium thiosulfate, and then add 1 mole and 2 mole concentrations of hydrochloric acid to heated and cooled sodium thiosulfate, and record the amount of time it takes to go white, or opaque. We know it is opaque because we have added a black cross underneath the beakers with the sodium thiosulfate. The time that we have recorded is called the 'reaction rate' The Collision theory explains how chemical reactions happen and why the rate of the reaction rates differ between different chemical reactions. It states that for a chemical reaction to occur, atoms, molecules, or chemical species have to collide in order to have a chemical reaction, but not all collisions bring chemical change. The Collision theory was proposed in 1916 by the German chemist Max Trautz, and British scientist William Lewis, in order to explain how chemical reactions and why rates differ for different reactions. In our experiment, we used different concentrations of hydrochloric acid, and in the Collision theory, lower concentrations have lower collisions, and higher concentrations have more collisions. That means that, theoretically, the higher concentrations of hydrochloric acid means that the 2 mole HCl will have more collisions, and that means that the reaction rate should be faster. Temperature is also a factor in the Collision theory. An increase in the temperature increases the average speed of the reactant molecules and the number or frequency of collisions. The Kinetic theory, states that the hotter the substance is, the faster the molecules move in it.
HYPOTHESIS
I believe that the heated sodium thiosulfate will react faster with the 2 mole hydrochloric acid. There are a number of variables to take into consideration, like the independent variable, which is that the temperature of the hot and cold thiosulfate could be different temperatures in the one mole and two mole experiments, the dependant variable is that the reaction rate could change. Six controlled variables would be the amount of thiosulfate and hydrochloric acid, the amount of icy water and hot water, the same stopwatches, if they were stirred or not, and they were done in the same climatic conditions. The purpose of a controlled group would be to see how the hot and cold thiosulfate differ from the normal room temperature thiosulfate, and then look at the difference between the 3 groups of experiments.
I believe that the heated sodium thiosulfate will react faster with the 2 mole hydrochloric acid. There are a number of variables to take into consideration, like the independent variable, which is that the temperature of the hot and cold thiosulfate could be different temperatures in the one mole and two mole experiments, the dependant variable is that the reaction rate could change. Six controlled variables would be the amount of thiosulfate and hydrochloric acid, the amount of icy water and hot water, the same stopwatches, if they were stirred or not, and they were done in the same climatic conditions. The purpose of a controlled group would be to see how the hot and cold thiosulfate differ from the normal room temperature thiosulfate, and then look at the difference between the 3 groups of experiments.
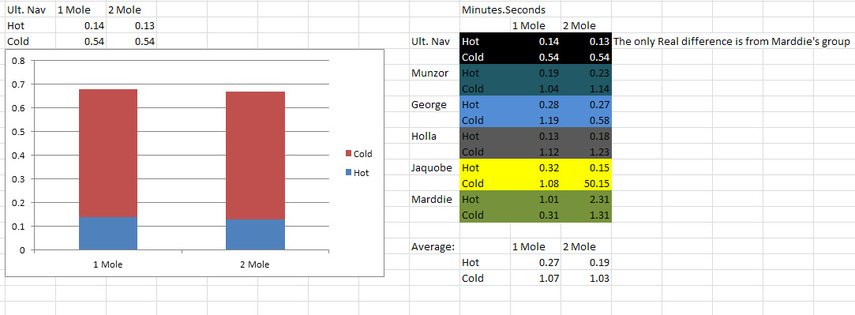
RESULTS
DISCUSSION
1. How does the temperature affect the reaction rate for this experiment?
The temperature affects the reaction rate because as the Collision theory states, the higher the temperature, the faster the molecules will vibrate, and that means that more collisions will happen, which means that the chemical reaction will happen faster. In most instances, the reaction was faster with the hot HCl than the cold HCl. The cold thiosulfate was slower because the molecules vibrated much slower, which meant the molecules didn't collide as often, so the chemical reaction was slower in most cases.
2. How does concentration of the hydrochloric acid affect the reaction rate?
The concentration affects the reaction rate again, because of the Collision theory, as it says that the higher the concentration, the more frequent the collisions happen, and if there are more collisions then in the 1 mole HCl, the reaction rate was faster in most cases. The aim of the experiment was to see how concentration affected the reaction rate, and according to the results, the difference
3. Can you be sure your answers in the above questions apply to all reactions?
Not really because most of the variables would change with different chemical reactions, for example you wouldn't be able to stir hot rust. Variables chaneg fr
1. How does the temperature affect the reaction rate for this experiment?
The temperature affects the reaction rate because as the Collision theory states, the higher the temperature, the faster the molecules will vibrate, and that means that more collisions will happen, which means that the chemical reaction will happen faster. In most instances, the reaction was faster with the hot HCl than the cold HCl. The cold thiosulfate was slower because the molecules vibrated much slower, which meant the molecules didn't collide as often, so the chemical reaction was slower in most cases.
2. How does concentration of the hydrochloric acid affect the reaction rate?
The concentration affects the reaction rate again, because of the Collision theory, as it says that the higher the concentration, the more frequent the collisions happen, and if there are more collisions then in the 1 mole HCl, the reaction rate was faster in most cases. The aim of the experiment was to see how concentration affected the reaction rate, and according to the results, the difference
3. Can you be sure your answers in the above questions apply to all reactions?
Not really because most of the variables would change with different chemical reactions, for example you wouldn't be able to stir hot rust. Variables chaneg fr
CONCLUSION
The fastest reaction rate was when the 2 mole HCl was added to the hot sodium thiosulfate. The main source of error was contamination. People weren't taking enough care when pouring and measuring the HCl and sodium thiosulfate, and in some instances, the reaction started happening and the experiment had to be restarted because the reaction was unintended. An easy way to resolve this error is to take more care when measuring and pouring the liquids, and cleaning up the chemical spills, as a lot of groups just left the HCl and sodium thiosulfate laying on the bench if they spilled it.
The fastest reaction rate was when the 2 mole HCl was added to the hot sodium thiosulfate. The main source of error was contamination. People weren't taking enough care when pouring and measuring the HCl and sodium thiosulfate, and in some instances, the reaction started happening and the experiment had to be restarted because the reaction was unintended. An easy way to resolve this error is to take more care when measuring and pouring the liquids, and cleaning up the chemical spills, as a lot of groups just left the HCl and sodium thiosulfate laying on the bench if they spilled it.