Elements
An element is a substance in its most basic form, it cannot be broken down any further. Plastic, paper and sugar, you may think they are elements, but they aren't, because they can all be broken down into simpler forms. An easy way to break down a substance is to burn it. If you set fire to any of the substances stated, it will leave black carbon, one of the elements from which it is made. The rest of the elements go up in smoke, steam, or a vapour of some sort. Carbon is an element, because you can't break it down any further. An example of some other elements are, gold, silver, copper, tin, oxygen, etc. There is a list of all the elements currently known to man called the Periodic Table of Elements, which is a separate section on this website.
There are 92 naturally occurring elements. There are over 20 synthetic elements: these have never been found by nature but actually created in laboratories. Many synthetic elements are unstable and only exist for a few seconds after being created.
There are 92 naturally occurring elements. There are over 20 synthetic elements: these have never been found by nature but actually created in laboratories. Many synthetic elements are unstable and only exist for a few seconds after being created.
Element Symbols

Each element has a unique symbol, so it can be identified. For example, hydrogen is H, helium is He, mercury is Hg, etc. H for hydrogen and He for helium seem normal, but why is mercury Hg? The Latin word for mercury is hydrargyrum, which means liquid silver. Many of the elements that have been known a long time get their symbols from Latin or Greek words.
Atoms
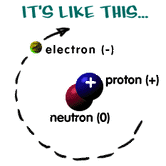
An atom is a basic unit that makes up all matter. There are many different types of atom each with its own name, mass and size. These are called chemical elements, such as hydrogen or gold. Atoms are very small. VERY small. About 0.1 to 0.5 NANOMETRES small! One nanometre is around a hundred times smaller than the width of a human hair. This makes atoms almost impossible to see. The way they work, and how they act with other atoms has to be seen using equations.The number of protons, neutrons and electrons an atom has determines what element it is. Hydrogen, for example, has one proton, no neutrons and one electron; the element sulphur has 16 protons, 16 neutrons and 16 electrons.
Compounds

A chemical compound is a chemical made by joining together atoms of different chemical elements. The different atoms are joined by chemical bonds. The elements stick together so strongly that the compound behaves like one substance. What chemical is made depends on what atoms it is made of and how they are joined together. Chemical structure of water Sodium chloride Chemical structure of sodium chloride Chemical compounds can be liquids, like water which is made from atoms of hydrogen and oxygen joining together. They can also be solids, like sodium chloride that contains sodium and chlorine atoms.
Mixtures

In chemistry, a mixture is a substance that is made up of two or more simpler substances. These substances can be chemical elements or compounds. A mixture can be a liquid, a solid, or a gas. A mixture is not the same as a compound, which is made of two or more atoms connected together.If one substance in a mixture dissolves in the other, it is called a solution. For example if sugar is put in water it forms a mixture, then dissolves to make a solution. If it did not dissolve, it would be called a suspension. Solids can be mixtures also. Many kinds of soil and rock are mixtures of different minerals. A mixture can be separated using a number of filtering methods like filtration and evaporation.
